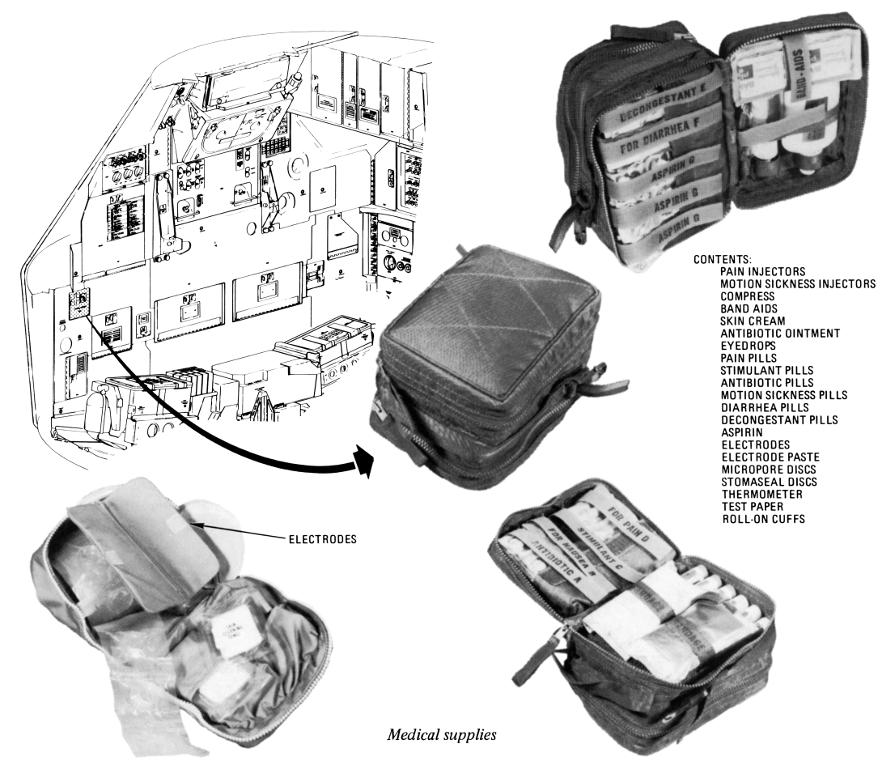
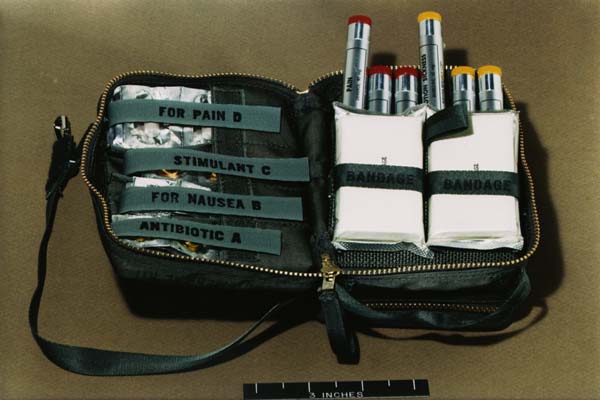
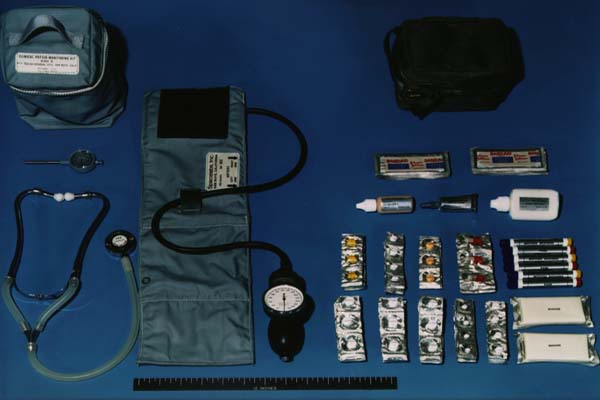
Apollo Medical Kit
The Apollo command module carried a small medical kit in the spacecraft's lower equipment bay. Stocked for contingency situations most likely to arise, it contained oral drugs and pills (for pain, motion sickness, and diarrhea; as well as stimulants, antibiotics, decongestants, and aspirin), injectable drugs (for pain and motion sickness), bandages, topical agents (first aid cream, sun cream, and an antibiotic ointment), and eye drops.
Click image for a 931x646 pixel version of this image in a new window.
From page 2-12-97 of the Apollo Operations
Handbook, Block II Spacecraft, Volume 1: Spacecraft Description;
refer to page 112 of the "Crew Personal Equipment" PDF.
Extraction and adaptation by heroicrelics.org.
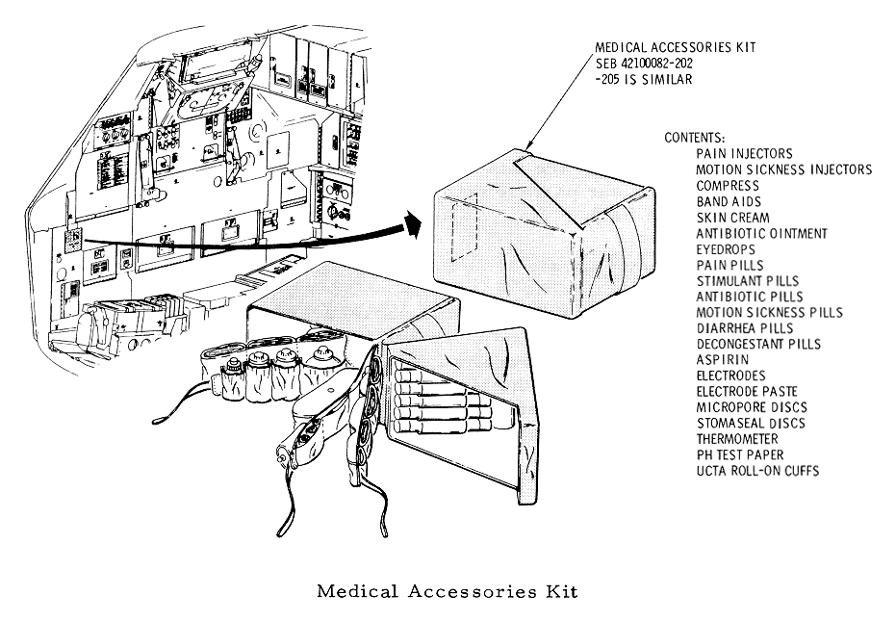
The medical kit was made of Betacloth and measured approximately 7" x 5" x 5".
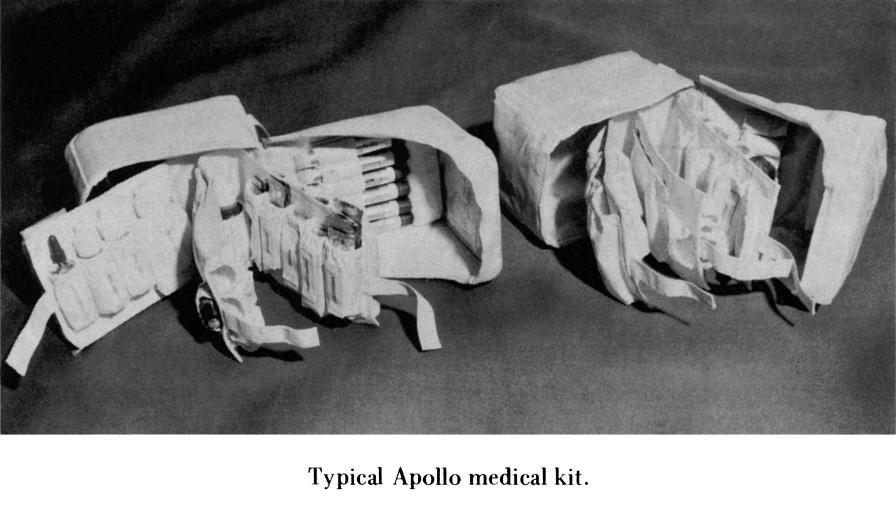
The Apollo medical kit, as stowed.
Click image for a 2268x2850 pixel version of this image in a new window.
Photo courtesy Historic Space Systems.
Opening the lid revealed three layers of medical supplies and equipment (a medical accessory kit with additional equipment was also stowed in a compartment behind the lunar module pilot's couch).
There was no "standard" medical kit throughout the program: While the basic contents of the kit remained largely the same from one mission to the next, the adequacy of the kit was reviewed after each mission and appropriate modifications were made for the next flight. A short-acting barbiturate, Seconal, was added after reports of sleep difficulties by the Apollo 7 crew. The cardiac arrhythmias experienced during the Apollo 15 mission resulted in the addition of Pronestyl, Lidocaine, atropine, and Demerol in subsequent missions.
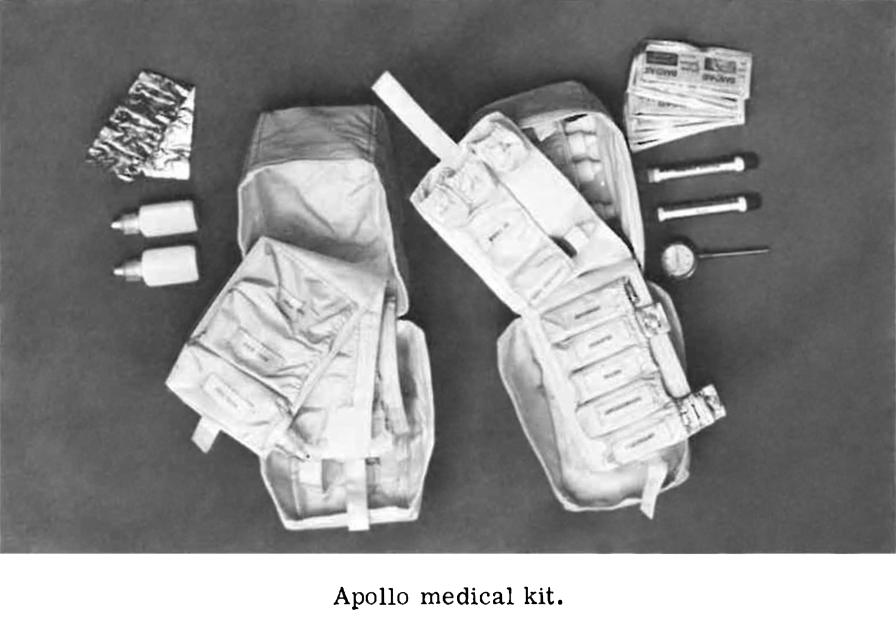
The Apollo medical kit, opened to reveal its contents.
Click image for a 2208x3102 pixel version of this image in a new window.
Photo courtesy Historic Space Systems.
The crew of Apollo 7 famously came down with colds, leading Apollo 7 commander Wally Schirra to later refer to the mission as the "11-day cold capsule". Twenty-four Actifed tablets were included in Apollo 7's medical kit, and all were consumed. The allotment of Actifed was increased to 60 pills for all subsequent missions. (Schirra went on to do a series of television commercials for Actifed in the 1980s.)
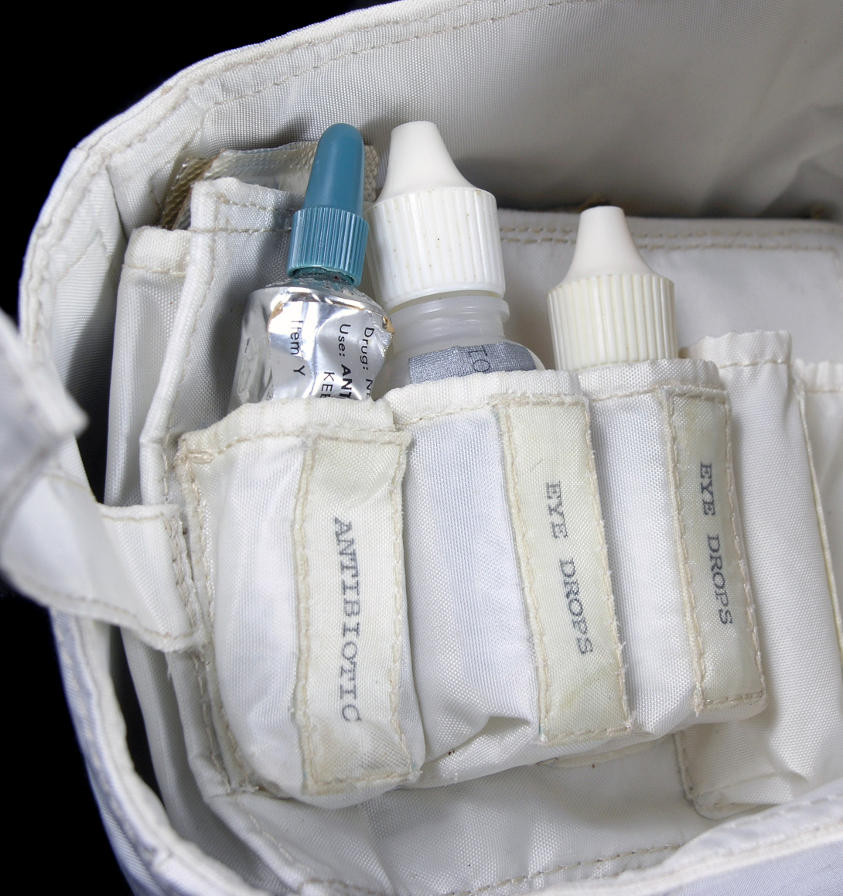
Detail of one of the flaps of the medical kit with a pouch designated for the
decongestant Actifed. A strip of tablets peeks out of the pouch.
Click image for a 2448x3264 pixel version of this image in a new window.
Photo courtesy Historic Space Systems.
The contents of the medical kits were also updated as more effective medications were identified. For example, the combination scopolamine/Dexedrine was substituted after Apollo 11 for the previously stowed Marezine after ground-based tests indicated it was more effective for the treatment of motion sickness.
All crewmembers were tested for both sensitivity and response to each of the medications carried in the medical kit. Tylenol and Benadryl were added to the medical kit for the Apollo 8 mission due the aspirin sensitivity of one of the crewman.
Click image for a 2558x1487 pixel version of this image in a new window.
From page 62 of Biomedical Results of Apollo, NASA SP-368, in the
heroicrelics library. Also available in electronic format from the NASA Technical Reports
Server.
Scan and cleanup by heroicrelics.
The Apollo medical kit evolved from the kit carried on Gemini missions. One of the objectives of the Gemini program was to conduct long-duration flights to verify that humans could live in space for the expected duration of a lunar flight. The duration of Gemini 7, for example, was 14 days, the duration of which made a medical kit a necessity. The Gemini 7 medical kit contained motion sickness tablets and injectors, stimulants, aspirin, pain pills and injectors, decongestants, anti-diarrhea tablets, antibiotic tablets, and eyedrops.
NASA's philosophy regarding the use of medication also evolved. Its initial policy precluded such usage except in a medical emergency (period documentation noted that "a normal man is preferred" and that "drugs are used only if necessary"). As additional experience was gained, this policy was relaxed and certain drugs were prescribed during Apollo missions when indicated. For example, sleeping pills were prescribed when adequate rest could not be obtained, particularly when sound sleep was important prior to critical mission phases.
Particular concern, however, centered on the ability of crewmen to perform effectively at periods of 1, 2, 3, and 4 hours following the ingestion of the Seconal sleeping aid. Crewmen were given flight-related performance tests at each of these four time intervals, and all exhibited satisfactory performance.
Note the Band-Aid brand adhesive bandages at upper left.
Click image for a 1640x1174 pixel version of this image in a new window.
From page 51 (page 59 in the PDF) of Apollo 7 to 11 -
Medical Concerns and Results.
Extraction and cleanup by heroicrelics.
Pills and tablets in the medical kit were packaged in such a manner that the crewmen had easy access to the medication at all times. The pills were sealed individually in cells in strips of 12 or 24 cells. Pressure-related problems in medication packaging were resolved by puncturing each cell with a small pin; the hole made it possible for the air to vent when under reduced-pressure conditions (the medical supplies would have been packaged at standard sea level pressures, but while in space the Apollo command module was pressurized to 5 psi).
More Band-Aids peek out from a pouch in the Apollo medical kit.
Click image for a 2444x2932 pixel version of this image in a new window.
Photo courtesy Historic Space Systems.
The use of standard spray bottles in a weightless, reduced-pressure environment proved unsatisfactory. Sprays were therefore replaced by dropper bottles, although these were not entirely satisfactory, either, as the likelihood of overdosage from a dropper bottle was increased.
Click image for a 2180x2316 pixel version of this image in a new window.
Photo courtesy Historic Space Systems.
An abbreviated version of the command module medical kit was carried in the lunar module. For the Apollo 11 mission, the lunar module kit contained eight Lomatil tablets, four Dexedrine tablets, 12 aspirin tablets, two Seconal capsules, one bottle of methylcellulose (1 percent) eye lotion, and two compression bandages.
The exact contents of the command module medical kit on each Apollo mission is contained in Table 12 on page 63 of the HTML version of the Biomedical Results of Apollo.
Although I found no specific information in this regard, it would appear that the medical kit was one of the items that was redesigned after the Apollo 1 fire. While all of the the photos of the as-flown medical kits showed that they were constructed of the familiar, white Betacloth as shown above, I found a number of photos and diagrams depicting a different configuration.
These first two photos appear to show largely the same contents to the medical kits as above, but the kit's enclosure is rather different.
NASA photo S66-28355.
The NASA IDs for these photos begin with "S66", indicating that they were issued in 1966, which would seem to explain the difference. However, I found these photos on a NASA Apollo 11-related page.
NASA photo S66-37423.
The following diagram is from an Apollo Spacecraft News Reference issued in preparation for the Apollo 11 mission. Oddly, it shows the same style of bag as the two photos immediately above. My first thought was that the News Ref was simply not updated from the Block I version of the publication, but I've scanned a Block I Apollo Spacecraft News Reference which contains only a few sentences (and no diagrams) about the medical kit.
Still, my best guess is that the photos above and this diagram reflect the Block I configuration of the medical kit.
Click image for a 4429x3844 pixel version of this image in a new window.
From p. 81 of the Apollo Spacecraft News Reference [Command/Service
Module], located in the Mrazek collection, Dept. of Archives/Special
Collections, M. Louis Salmon Library, University of Alabama in
Huntsville. A similar-vintage News Reference is available in
electronic format from the Apollo Lunar
Surface Journal; refer to page 85 of that PDF.
Scan and cleanup by heroicrelics.
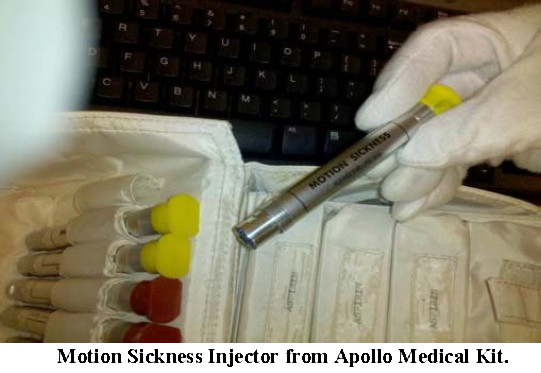
One of the more interesting types of items in the medical kit was the medical injector. Injectors were provided to deliver drugs for motion sickness (yellow cap) and for pain (red).
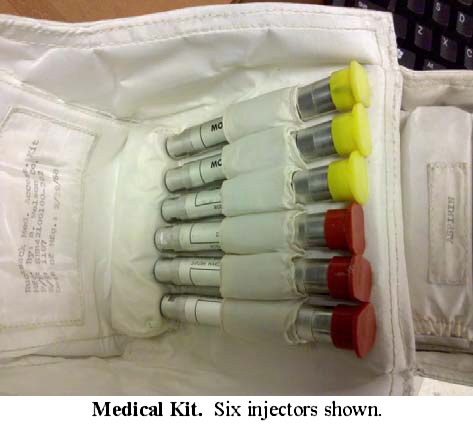
From page 7 of "Breaking the Pressure
Barrier: A History of the Spacesuit Injection Patch".
Extraction and cleanup by heroicrelics.
From page 7 of "Breaking the Pressure
Barrier: A History of the Spacesuit Injection Patch".
Extraction and cleanup by heroicrelics.
Mission planners identified the need to administer medication to a crewmember during a mission in the event of illness at the very beginning of the Apollo program. In a pressurized cabin, it would have been possible to administer medicine, via a pill or an injection, as would normally be done on Earth. However, during a contingency, the crew member might require medication during an extravehicular activity on the lunar surface or in an unpressurized spacecraft (e.g., if hull integrity of the command module were compromised). In such a situation, the astronaut would be wearing a pressurized suit, making administering medication more problematic.
The Apollo space suit was thus designed to have a biomedical injection patch to facilitate an injection in such circumstances. The Apollo Space Suit Assembly Design and Performance Specification, October 12, 1964, imposed the following requirement:
Medical Injection Provisions - The PGA [pressure garment assembly] shall provide the capability for the crewman to administer to himself hypodermic injections utilizing a spring-loaded plunger type needle, while in a pressurized PGA. The PGA shall provide, in a location to be determined by NASA, features which shall allow the insertion of the needle and subsequent withdrawal, without endangering the pressure integrity or reliability of the suit, and shall be self-sealing to prevent the loss of gas at the site of the needle penetration. Best location for medical injections is on the ventrolateral aspect of the thigh, approximately half-way between the knee and the hip. An alternate location would be the deltoid area of either arm.
The injection patches were sewn into the PGA's restraint layer using a zigzag pattern, providing visual confirmation of the injection site, and were integrated between the restraint and pressure bladder. When the patch was to be used, the needle would puncture the restraint, then the injection disc, then the bladder before entering the suit cavity.

Full-sized crop from my Apollo A7L Suit TLSA and ITMG photo set at the Franklin Institute showing the biomedical injection area (red stitching at left).
To integrate the thermal micrometeorite garment (TMG), a large "biomedical injection flap" was added. This flap was kept in place by buttons and Velcro. Opening the flap allowed access to both the injection patch and the urine transfer connector (UTC).
Tubing in the liquid cooling garment (LCG) was strategically routed away from the injection site on the right thigh to avoid puncturing the water lines running through the LCG.
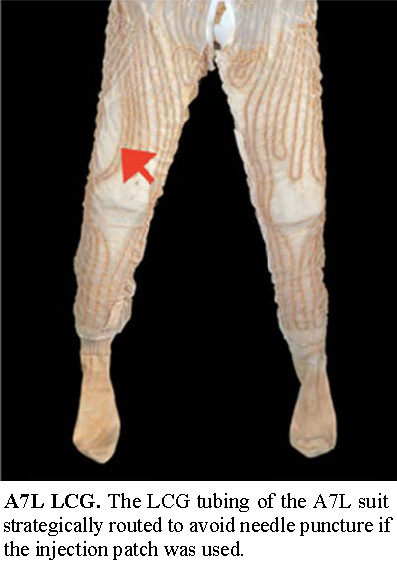
Adapted from page 5 of "Breaking the Pressure
Barrier: A History of the Spacesuit Injection Patch".
Extraction and adaptation by heroicrelics.
The injection patch was relocated to the left thigh in the A7LB suit. This was a result of the cascade of changes initiated by the relocation of the suit's pressure gauge. It was originally located on the right wrist of the A7L but was relocated to the left wrist for the A7LB. This forced the pressure relief valve (originally on the left wrist of the A7L) down to the injection patch location on the right thigh and, finally, the injection patch was moved from the right leg to the corresponding location on the left thigh.

Full-sized crop from my Kerwin Skylab Suit photo set at the U.S. Space & Rocket Center, showing the biomedical injection patch on the left leg of the suit.
Apparently, little documentation exists regarding the composition of the injection patch, so for a paper presented in 2013, researchers from Johnson Space Center and Glenn Research Center removed the injection patch from Frank Borman's Apollo 8 A7L suit. Non-destructive testing revealed that the patch consisted of two sides with slightly different materials. The patch was over 97% silicone, with large traces of clorine and titanium and smaller traces of various other elements.

Adapted from page 6 of "Breaking the Pressure
Barrier: A History of the Spacesuit Injection Patch".
Extraction and adaptation by heroicrelics.
Specific details on the engineering design of the Apollo medical injector are also scarce; the researchers were unable to locate engineering drawings, specifications, requirements, or internal pictures of the devices. However, they did interview a doctor who served as a flight surgeon during the Apollo program.
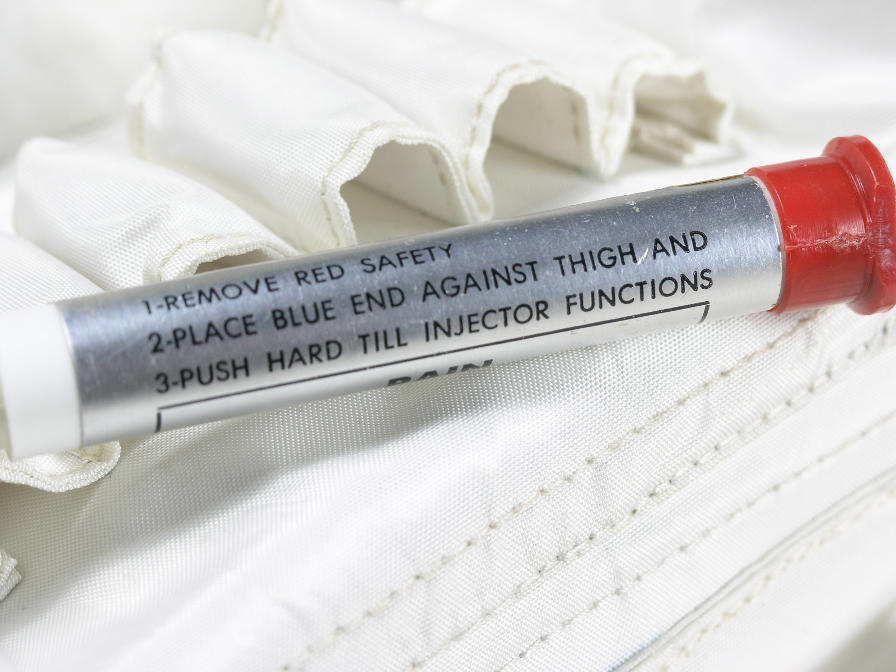
From his description, the injectors housed pre-filled medical syringes inside a pressurized aluminum tube. This tube would be placed flush with the outside of the restraint layer and activated by pressing a button at the top of the device. Once the button was depressed, a spring performed two actions: First, it displaced the syringe and needle, driving the needle through the seal on the injector, through the injection patch in the suit, and into the muscle on the upper thigh. The spring continued its action to depress the plunger on the syringe, delivering the medication into the muscle of the crewmember. The time from depressing the button on the injector to complete delivery of the medication was fast (less than 1 second) to prevent the crewmember being injected from reflexively pulling away before the injection was finished.
Directions for use on an Apollo medical injector.
Click image for a 3264x2448 pixel version of this image in a new window.
Photo courtesy Historic Space Systems.
The bulk of the descriptive text on this page was taken, largely verbatim, from the Biomedical Results of Apollo (NASA SP-368), Apollo 7 to 11 - Medical Concerns and Results, and "Breaking the Pressure Barrier: A History of the Spacesuit Injection Patch".